Mouse study reveals how chronic stress promotes breast cancer stem cells, identifies vitamin C as effective therapy
URBANA, Ill. – Cancer: The word alone evokes dread, anxiety, and fear. Accordingly, many women living with the disease and undergoing treatment experience chronic stress and depression. Scientists have demonstrated, in studies with rodents and humans, that stress can exacerbate cancer’s progression, but it wasn’t clear how.
A new study, published in The Journal of Clinical Investigation, establishes that the stress hormone epinephrine sets off a cascade of biochemical reactions that favor breast cancer growth and spread.
In the study, the researchers first demonstrated the effects of chronic stress on cancer stem cell growth, a novel twist on previous research that did not specifically focus on these self-perpetuating cells.
“You can kill all the cells you want in a tumor, but if the stem cells, or mother cells, are not killed, then the tumor is going to grow and metastasize. This is one of the first studies to link chronic stress specifically with the growth of breast cancer stem cells,” says Keith Kelley, emeritus professor in the Department of Animal Sciences and the College of Medicine at the University of Illinois, and an author on the study.
To do this, they induced chronic stress in mice, by placing them in small enclosures that limited their movement. All the mice were stressed for a week before being inoculated with either human or mouse breast cancer cells. After inoculation, the mice were split into two groups: controls, which were moved into large cages; and stressed, which stayed in the small enclosures for an additional 30 days.
Confirming the researchers’ expectations, the mice experiencing chronic stress showed behavioral changes consistent with anxiety and depression. They also had bigger, faster-growing tumors and more cancer stem cells than mice in control conditions.
Having demonstrated the link between chronic stress, mood changes, and enhanced growth of breast cancer stem cells, the scientists went on to investigate the underlying biochemical underpinnings that caused stress to increase growth of cancer cells.
“The direct signaling network between stress pathways and a cancer-propagating system remains almost completely unknown,” says Quentin Liu of the Institute of Cancer Stem Cell at Dalian Medical University in China and principal investigator on the study. “A better understanding of the biochemistry that causes stress to increase the growth of cancer cells could lead us toward targeted drug interventions, one of which we discovered in this work.”
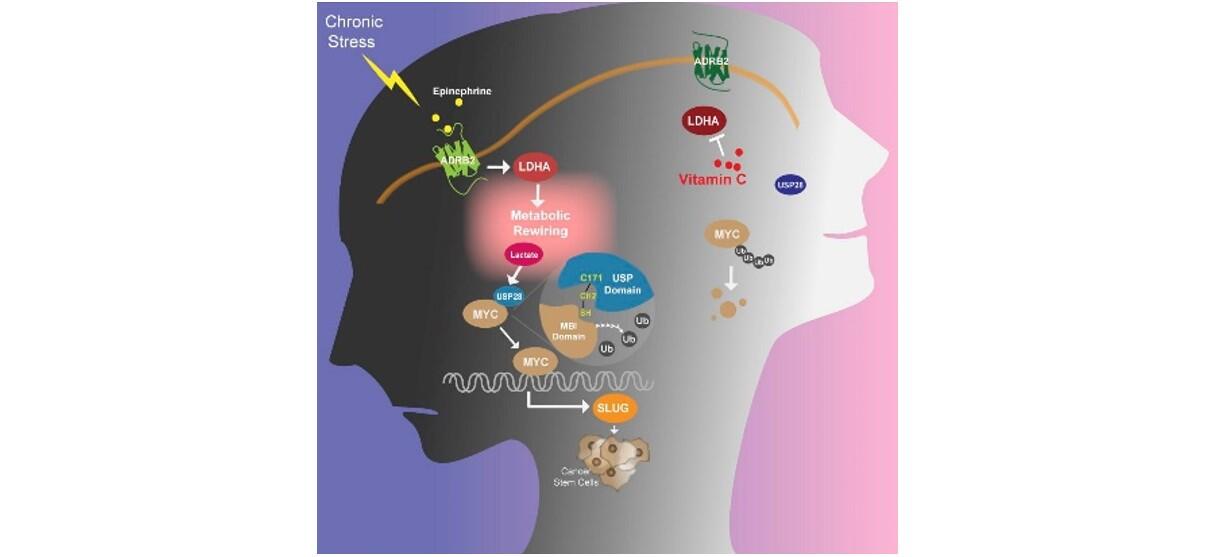
Multiple lines of evidence led the scientists toward epinephrine, one of the body’s major stress hormones. First, epinephrine levels were significantly elevated in mice that experienced stress for the duration of the experiment. Second, in stressed mice that received treatments to inactivate the receptor for epinephrine – ADRB2 – tumors were significantly smaller and fewer stem cells were found.
“When most people think of stress, they think it’s cortisol that’s suppressing the immune system. The amazing thing is cortisol was actually lower after a month of stress,” Kelley says.
Once epinephrine binds to one of its two receptors, ADRB2, it elevates levels of an enzyme called lactate dehydrogenase. In normal situations, this enzyme delivers quick energy to muscles in a fight-or-flight situation and produces lactate as a byproduct. But cancer cells need lactate for energy. With excessive amounts of lactate dehydrogenase in chronically stressed individuals, cancer-causing genes are activated and cancer cells proliferate.
“These data provide a novel pathway that explains how elevated epinephrine caused by chronic stress promotes breast cancer progression by acting directly on cancer stem cells,” Liu says.
To evaluate the clinical significance of their findings with mice, the scientists measured epinephrine in the blood of 83 human breast cancer patients. Women with high levels of the stress hormone also had high levels of lactate dehydrogenase in biopsied breast cancer tissue, compared with adjacent non-cancerous tissue. Importantly, and consistent with findings in mice, patients with high serum epinephrine had significantly lower overall survival and disease-free survival compared to patients with low epinephrine levels.
In a final test, the researchers grew breast cancer cells in the lab and introduced a wide variety of FDA-approved cancer drugs. Several treatments, including vitamin C, suppressed lactate dehydrogenase production. When vitamin C was injected into stressed mice, tumors shrank.
Scientists have suspected Vitamin C’s cancer-fighting potential for decades, and several clinical trials have demonstrated positive results. This study contributes a new understanding of the vitamin’s action in biochemical pathways relevant to chronically stressed breast cancer patients.
“Taken together, these findings show that vitamin C might be a novel and effective therapeutic agent for targeting cancer in patients undergoing chronic stress,” Liu says.
The article, “Stress-induced epinephrine enhances lactate dehydrogenase A and promotes breast cancer stem-like cells,” is published in The Journal of Clinical Investigation [DOI: 10.1172/JCI121685]. Authors include Bai Cui, Yuanyuan Luo, Pengfei Tian, Fei Peng, Jinxin Lu, Yongliang Yang, Qitong Su, Bing Liu, Jiachuan Yu, Xi Luo, Liu Yin, Wei Cheng, Fan An, Bin He, Dapeng Liang, Sijin Wu, Peng Chu, Luyao Song, Xinyu Liu, Huandong Luo, Jie Xu, Yujia Pan, Yang Wang, Dangsheng Li, Peng Huang, Qingkai Yang, Lingqiang Zhang, Binhua P. Zhou, Suling Liu, Guowang Xu, Eric W.-F. Lam, Keith W. Kelley, and Quentin Liu. The research was supported by the National Natural Science Foundation of China; the Science and Technology Planning Project of Guangzhou; and the Dalian High-level Talent Innovation Program.